A UNIQUE OUTDOOR EDUCATION AND ADVENTURE EXPERIENCE IN JULY ON HORNBY ISLAND
for more information, click here
for more information, click here
THe transfer of energy and nutrients through ecosystems
|
Topics for Chapter 2 Energy and Nutrients through Ecosystems
|
|
|
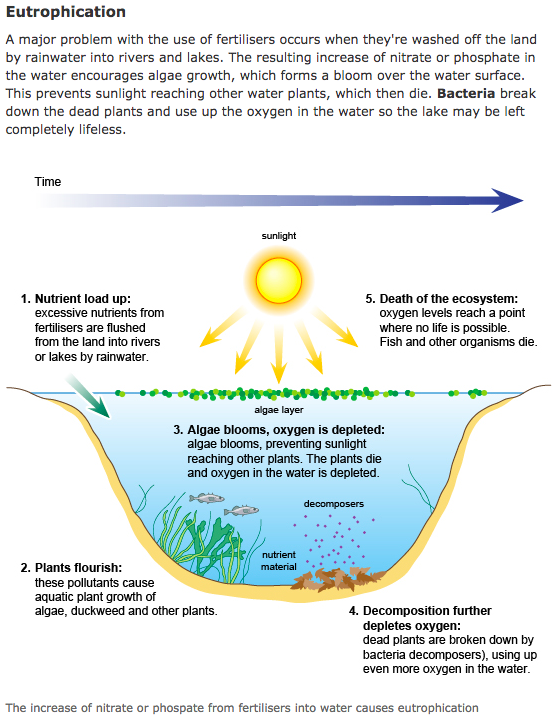
A thank you to GCSE Bitesize for the information provided to the left. For more science information, click on the image to be directed to GCSE Bitesize website.
June 14, 2018
Section 2.2 from BC Science 10, pages 68 to 91 and page 232, and page 105
Begin Heavy Metal Discussion/Bioaccumulation June 13, 2018
|
|
|
June 6th, 2018
|
June 5, 2018
- Notes on Ecosystems: Food Webs and Energy Transfer. Link to website
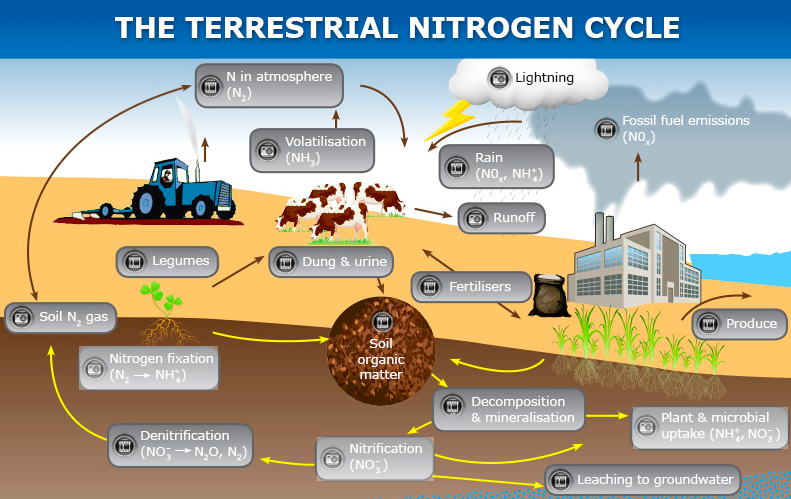
- Notes on Carbon
- Article, Interactive on the Carbon Cycle - Read and view to answer the following questions LINK to WEBSITE
- List and describe the processes on how living things get carbon.
- List and describe how carbon is returned to an inorganic state. Watch videos within website.
- Define the term emission. After watching the brief video, list and describe the processes which emit carbon into the atmosphere.
- Describe two process within the ocean that allow for the take up and storage of carbon within the ocean.
Energy and Matter Transformation
May 22, 2018
Answer questions on page 33 #3-8
May 18, 2018
Presentation of adaptations of native plants of Canada, due to the factors which limit photosynthesis - obtaining energy
Copy notes from someone who was in class Friday, as you will be responsible for this information on your assessment.
Assignment: Continued from yesterday: page 52, #1-4 AND #5-7, answers for 5-7 on pages 18 - 19 and 23.
Answer questions on page 33 #3-8
May 18, 2018
Presentation of adaptations of native plants of Canada, due to the factors which limit photosynthesis - obtaining energy
Copy notes from someone who was in class Friday, as you will be responsible for this information on your assessment.
Assignment: Continued from yesterday: page 52, #1-4 AND #5-7, answers for 5-7 on pages 18 - 19 and 23.
|
May 16, 2018
|
May 17, 2018 Introduction to adaptations - watch Primeval New Zealand - link below,
|
|
FREE WRITE: to write continuously for 10 minutes without stopping, the intent is to get a flow of ideas onto paper without worrying about the quality of the writing, but on the quantity
|
|
|
May 15, 2018
|
|
|
May 14, 2018
|
Difference between Potential and Kinetic Energy
|
Unit: Electricity
Current Electricity - CH 10: Science Probe 9
|
This includes the following assignments:
|
As well as the following labs:
|
|
May 15, 2018 - Chapter 10: Quiz Current Electricity
May 7, Return of Practice Test - answer questions, introduce next chapter May 3, 2018 - Practice Test - Unit Review and Kahoot May 2, 2018
|
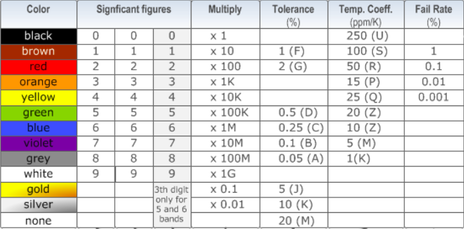
RESISTOR COLOR CODE CHART
|
|
Friday April 27, 2018
After you have ensured past due assignments listed below have been completed, start today's assignment, if completed today you will not have to complete the assignment at home over the weekend.
|
Thursday April 26,2018
If Activity is not finished - it must be completed by the end of the class today (or at least the iPad part of the activity) Assignment: Page 309 #1-6, 8 & 9 |
|
Wednesday April 25, 2018
|
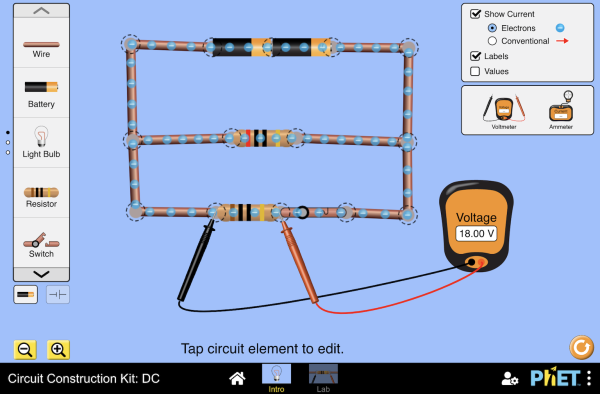
Review of Series and Parallel Circuits below:
|
Friday April 20, 2018
Obtain Ch 10 Booklet from Ms Gambrel
Obtain Ch 10 Booklet from Ms Gambrel
- Refer to the Vocabulary list at the end of the Chapter - In Cornell Note Format or Flash Card Format, define the words on the list up to Electron Flow,
- Answer Questions #1-5 at the end of section 9.1
STATIC ELECTRICITY - CH 9: SCIENCE PROBE 9
|
Thursday April 19, 2018: Chapter 9 Quiz on Static Electricity
Tuesday April 17: Practice Test and Kahoot for Review for Thursday's Quiz Monday April 16: Hand in page 281 #1-10, Page 278 #1-12 and Activity 9B: The Electroscope (all past due) and Page 284 #1-7, assigned this past Friday. Review of Lightning - video: 10 minute free write on lightning Wednesday Assignment: Friday April 13: Lab Corrections for Activity 9B: The Electroscope must be submitted prior to the end of class Review Vocabulary over the weekend, for Thursday's quiz Thursday April 12: Vocabulary for Chapter 9: to be completed by the end of class, list of vocabulary words are on page 294 of Ch 9: Static Electricity handout. Finish watching video on Lightning - note taking Wednesday April 11: Finish first copy of activity 9B: The electroscope Begin watching video - "Raging Planet - Lightning" - take notes on video for reference in 10 minute free write |
Tuesday April 10:
Monday April 9: Friday April 6 Thursday April 5 Wednesday April 4: Try this activity on page: the pith ball - hand in activity sheet Assignment: page 278 #1-7 Introduction to static electricity |
March 12, 2018
Complete Mind Map of Chemistry/Matter
Complete in Flash card format or Cornell note format:
Page 113 # 5, 6, 10, 11, 12, 13, 16
February 28, 2018
February 27, 2018
WEEK 4
February 21, 2018
Notes on naming and writing the formulas of ionic compounds in which the metal is multivalent, i.e. has more than one combining capacity.
February 14, 208
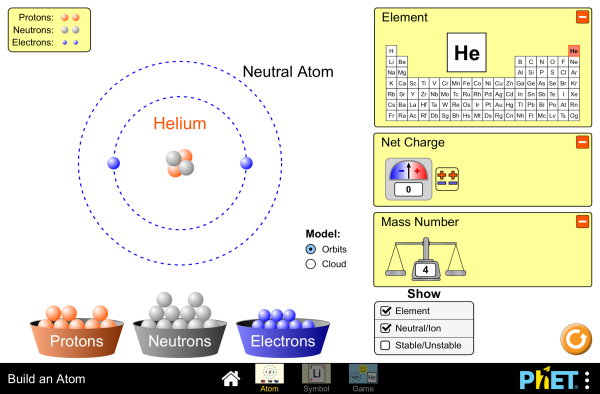
Working to understand atomic structures of elements - using the periodic table: number of protons, neutron and electrons of neutral atoms as well as ions.
February 13, 2018
You were given time on Friday to complete the following and what was not completed was to be completed at home over the weekend. If it is not done by the end of class today, as well the handout assigned today. If you did not finish, I should see you in Flex on Wednesday February 14.
February 9, 2018
Complete Mind Map of Chemistry/Matter
Complete in Flash card format or Cornell note format:
Page 113 # 5, 6, 10, 11, 12, 13, 16
February 28, 2018
- Individual handout to complete on writing chemical formulas - quiz on Friday in regards to this topic
- metals with or without a multivalency and a nonmetal
- metals with or without a multivalency and a polyatomic
- ammonium and a nonmetal
- ammonium and a polyatomic
February 27, 2018
- Reviewed how to write chemical formulas, particularly using multivalent metals
WEEK 4
February 21, 2018
- Notes: Covalent vs Ionic, introducing Polyatomic Ions (molecules which have an ionic charge)
- Assignment: Handout:
- Warm-up (Practice for tomorrow's quiz):
- Textbook:
- Pg 86 #1 a,c,e,i,l
- Pg 87 #1 a,b,d,f and #2 a,c,e,g,h
- Pg 89 #1 a,b,g,h,j,k
- Pg 90 #1 a,c,e,g,i,k,m
- Textbook:
Notes on naming and writing the formulas of ionic compounds in which the metal is multivalent, i.e. has more than one combining capacity.
- assignment - finish yesterday's assignment and hand in,
- complete today's assignment - if not completed, finish as homework or come in during flex tomorrow to complete.
- Reviewed: writing chemical formulas of ionic compounds where the metal has only one combining capacity
- Introduced nomenclature in how to properly write the name of ionic compounds
- In class, complete questions from the textbook on page 83 #1-5
- Complete page 46 of handout, if not finished in class, complete as homework.
February 14, 208
Working to understand atomic structures of elements - using the periodic table: number of protons, neutron and electrons of neutral atoms as well as ions.
February 13, 2018
You were given time on Friday to complete the following and what was not completed was to be completed at home over the weekend. If it is not done by the end of class today, as well the handout assigned today. If you did not finish, I should see you in Flex on Wednesday February 14.
- Complete Periodic Table (back) - for each element the following should be included:
- Neutral Lewis Dot Structure,
- Lewis Dot structure for ion,
- number of protons (atomic number) should be recorded in the upper left hand corner,
February 9, 2018
- Complete Handout on Characteristics of Families
- Video on the six Alkali metals and their characteristics, click here
- Complete (homework if not completed in class), the back side of the Periodic Table - which for each element you should have the Lewis Dot Structure for the neutral atom and for the ion.
|
|
|
Make sure the following have been handed in for credit:
|
February 8, 2018
- Spectroscopes: activity to be completed in the classroom
- You were assigned the following questions to answer at the bottom of your Periodic Table:
- As you move down the Families (columns) on the Periodic Table:
- List similarities between the elements in regards to the Bohr and Lewis Dot Structures, this answer should be the same for each family on your Periodic tables,
- what do you observe about the shells as you move down the rows?
- As you move across the rows (left to right), what do you observe about the arrangement of electrons? again answer should fit each row in your Periodic Table
- As you move down the Families (columns) on the Periodic Table:
|
February 6, 2018
|
February 5, 2018
February 2, 2018 - to be handed in by end of class: everything from Wednesday, Thursday and today
- View and take notes: Chemistry: 100 Greatest Discoveries in Science with Bill Nye, click here for link,
- start at 17 min and view to 29:30, starts with Periodic Table to Chemica Bonds
- Due in class today:
- States of Matter Handout,
- Cornell Notes,
- Atomic Structure Handout
February 2, 2018 - to be handed in by end of class: everything from Wednesday, Thursday and today
- Cornell Notes: from BC Science 9 pages 28 to 33
- Completion of Handout: Atomic Structure
- Discussion of assignment: What is Matter?
- Video: Chemistry: 100 Greatest Discoveries in Science with Bill Nye, click here for link
- 10 Minute Free Write on Video
- Activity: What is a Chemical? What is not a Chemical?
- Discussion
- Work on assignment: What is Matter?
- Difference between Inference and Observation using an image